Home » lead acid battery » What causes lead acid battery to fail?
What causes lead acid battery to fail?

At present, the batteries that have been widely used include: lead acid batteries, nickel metal hydride batteries, nickel metal chloride batteries, sodium sulfur batteries, sodium chloride batteries, lithium ion battery and other commercial chemical batteries.
This article will introduce lead acid battery from several aspects.
- The composition of lead acid battery
- How does a lead acid battery charge and discharge?
- New type lead acid battery
- How is the recovery rate of lead acid battery?
- Is there a standard for the size of lead acid battery?
- Do lead acid battery go bad?
- The new advances of lead acid battery
- Organizations about lead acid battery
Lead acid battery, named after the anode and cathode of lead plates and sulfuric acid electrolyte, is the oldest and most common type of secondary battery, and have been widely used in energy storage. Although lead acid batteries are reliable and stable, they have a low cycle life (about 300 to 500 cycles).
Lead acid battery is the most reliable and cheapest power supply system in the early days, and found some application in electric vehicles and it is still the most common backup power source. Because of its high power performance, lead acid batteries are mainly used in engine starting, lighting, ignition and so on. In the future, due to the relatively low price, lead acid batteries will still occupy a certain market share.
The composition of lead acid battery
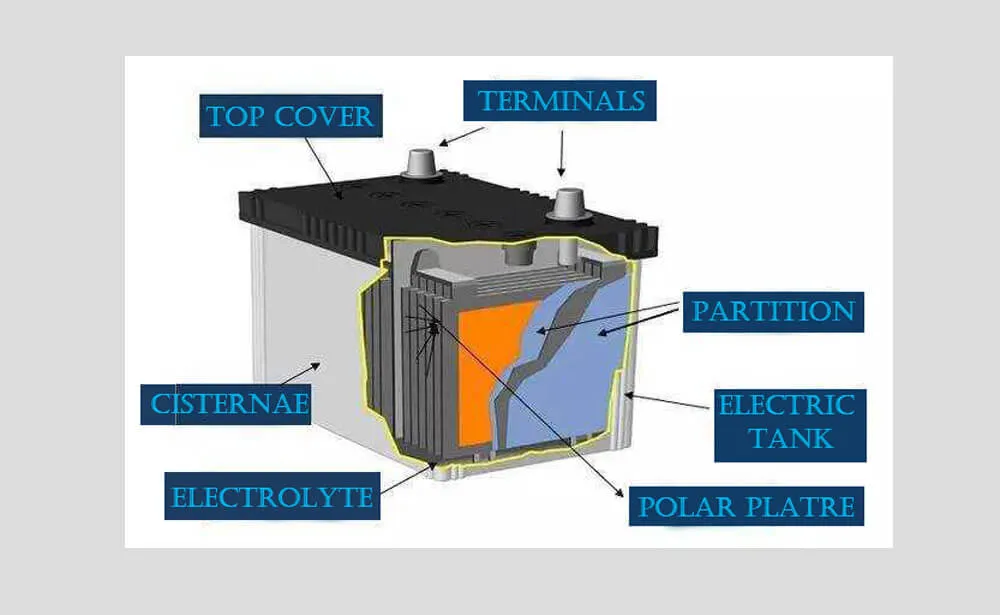
In terms of composition, lead acid battery mainly consist of lead and its oxides and a paste like lead plate or lead net composed of sulfuric acid as the electrode. These lead plates are connected to each other and immersed in a solution of sulfuric acid. The positive lead disk is mainly composed of lead dioxide, while the negative electrode is composed of spongy lead.
At present, most of the lead plates are added with jin, tin, calcium, selenium and other elements to form alloys. Alloyed lead plates can improve the rigidity, which is beneficial to the battery preparation process and the final product quality. The most common diaphragm between positive and negative poles is a microporous plastic film. The number of lead disks is usually even, and the positive and negative disks are individually connected to each other, as shown in Figure.
How does a lead acid battery charge and discharge?
The charge and discharge process is as follows : During discharge, lead absorbs sulphate from the electrolyte, while the charge process releases sulphate into the electrolyte. In this process, the active material falls from the lead plate and the collector fluid to the bottom of the battery, thus limiting the cycle life of traditional lead acid batteries.
For this reason, most lead acid batteries are designed to leave a space at the bottom of the battery between the falling active material and the fluid collector to avoid short circuit contact. In addition, the reaction of lead and acid will produce a certain amount of gas in the electrolyte, so it is necessary to frequently check or replace the electrolyte of lead acid battery.
New type lead acid battery
Recently, a new type of lead acid battery has been commercialized. Unlike traditional lead acid batteries, the new battery uses a sponge of fiberglass cloth as a diaphragm that holds the electrolyte, rather than immersing the lead disk directly in the acid solution. This reduces leakage of the electrolyte and absorbs the gas from the reaction of the lead plate with the acid inside the diaphragm.
As a result, the battery does not require follow up services such as electrolyte inspection and reinjection after assembly. Such a battery can be classified as a valve controlled sealed lead acid battery, which has the main advantage that the gas produced by the battery can be returned to the electrolyte after the battery is closed. For these reasons, they are called maintenance free batteries.
How is the recovery rate of lead acid battery?
Although lead and sulfuric acid are hazardous substances in lead acid batteries, high recovery greatly reduces this disadvantage.
As the battery product with the highest recovery rate, the lead acid battery recovery rate in the US market is as high as 95% 98%, but this still does not meet the requirements of environmental protection, because the release of 2% 5% lead will still cause great harm to the environment.
In this regard, people hope to continue to improve the recovery rate of lead acid batteries.
Is there a standard for the size of lead acid battery?
In addition to environmental concerns, the current confusion over the size of lead acid batteries is another issue that is often debated. Although lead acid batteries have been standardized and commercialized on a large scale, different applications have different requirements for lead acid batteries, resulting in the dry variation of size standards.
The Battery Council International (BCI) has tried to set standards for the size of lead acid batteries. In addition to size standards, BCI has also developed a series of standards for lead acid batteries, such as operating voltage and terminal configuration.
Do lead acid battery go bad?
Lead acid batteries still have a high market share due to low material and manufacturing costs and high recovery rates. Traditional and valve controlled sealed lead acid batteries were initially used as starting batteries for motor vehicles. In recent years, lead acid batteries have developed rapidly in the field of electric bicycles and occupy an absolute dominant market share.
In addition, lead acid batteries are still a good choice for backup power sources, stationary energy storage and elevator applications that require high power but low battery cycle and specific energy requirements.
The new advances of lead acid battery
While lead acid batteries have been slow in development over the past 150 years, there have been some developments that have made them suitable for new types of electrification engineering, such as microhybrids and hybrids. Companies such as Energy Power System (EPS) and Ecoult have developed advanced PDA systems. EPS has developed a planar layered matrix for lead acid battery, which increases the battery life by two or three times and greatly improves the charging rate.
The performance has been close to the nickel metal hydride battery, but the price is about 1/3 of the nickel metal hydride battery. The new lead acid batteries are expected to be used in microhybrid and hybrid electric vehicles, as well as in power storage systems. The advanced process developed by EPS makes lead acid batteries more economically advantageous in market applications.
Ecoult, an Australian company, has developed the superlead acid battery, which is a modification of conventional lead acid batteries. The introduction of a carbon based electrode in a hyperlead acid battery is equivalent to the introduction of a supercapacitor in a lead acid battery. Superlead acid batteries, which can achieve thousands of cycles without significant electrochemical performance degradation, have been widely used in renewable energy, power grids, and hybrid electric vehicles.
EPS and Ecoult have made great progress in lead acid batteries in recent years, and they believe that lead acid batteries still have a lot of potential. They believe that time will prove that lead acid battery applications in microhybrid, hybrid, national grid and energy storage are a very good solution economically and pragmically.
Organizations about lead acid battery
Currently, there are many vendors, companies and organizations offering lead acid batteries for different applications. Founded in 1925, THE International Battery Council (BC) is a non profit organization that mainly leads different lead acid battery manufacturers to formulate standards for lead acid batteries, collect industry data, and promote information exchange among more than 200 international manufacturers.
Another important organization is the Advanced Lead Acid Battery Council (ALABC), founded in 1992, which is an international RESEARCH and development alliance focused on improving the specific capacity of Lead Acid batteries. Although ALABC is smaller than BCI, Only more than 70 members, but in improving the performance of lead acid battery research and development has an irreplaceable role.