Are the non-rechargeable dry cell battery still in use

What is a dry cell battery
The features mentioned above is relative to a battery that has a flowable electrolyte, as it does not contain any free liquids, making it best for portable devices that are often used as flashlight batteries and used a power source for cameras, radios, etc. Common dry batteries are zinc-manganese batteries, generally 1.55~1.75V, the nominal voltage is 1.5V.
The zinc-manganese battery is a primary battery with manganese dioxide as the positive electrode, zinc as the negative electrode, and ammonium chloride aqueous solution as the main electrolyte.The size models are mainly R20, R14, R6 and R03. Other dry batteries are mainly aa and aaa batteries (for more information, please see aa vs aaa battery).
Is dry cell battery battery rechargeable
A dry cell battery cannot be recharged because it is a type of battery cell designed to be used just once and then discarded. They cannot be recharged with electricity and reused like rechargeable batteries.
The reason they can’t be recharged is due to the fact that the dry cell battery contains electrolytes that are in a paste, and the more the dry cell battery is used, the chemicals that generate power are being used up by the chemical reactions are irreversible in the battery, with no charging system and the moment they are gone completely, the battery becomes useless as it will no longer produce electricity.
Only rechargeable batteries can be recharged and reused multiple times. They are also known as secondary cells, and examples include Lithium-ion cells.
Which is better-dry cell battery or wet cell battery
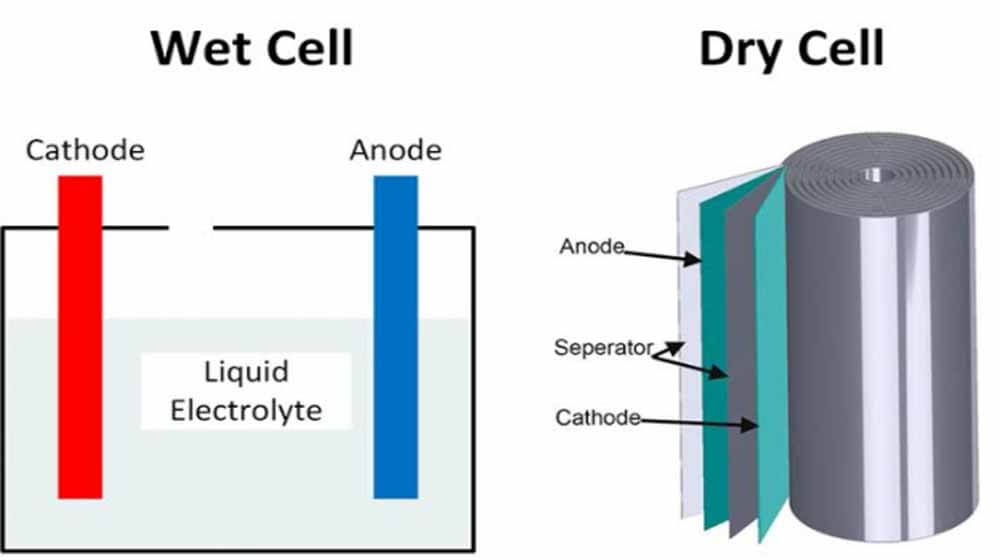
The wet cell battery also known as the original type of rechargeable battery contains a liquid electrolyte and is commonly found in cellphone towers, electric utilities and energy storage.
Commonly available and very cheap, the downside to the batteries is the required maintenance needed to maintain the liquid electrolyte it contains to a recommended and required level as insufficient electrolyte level leads to reduce performance, and you will need to recharge the battery.
Differences between the wet cell battery and the dry cell battery:
- Dry batteries are one-time use and cannot be recharged, while wet batteries are generally lead-acid batteries that can be recharged and used again.
- The electrolytes of a wet cell battery needs to be periodically checked and maintained, while no maintenance is required for the dry cell battery.
- The wet cell battery produces gases that are dangerous to human health, while the dry cell battery does not emit gases.
- The wet cell battery can only be used only when placed in an upright direction; any other direction may result in the spilling of acid, while the dry cell battery can be used in any direction without it spilling.
- The wet cell contains a liquid electrolyte and is heavy in weight, while the dry cell is relatively lighter and contains a paste electrolyte.
The dry cell battery is the better of the two since they are considered environmentally friendly, Because the country is now mercury-free and cadmium-free for alkaline dry batteries is the primary requirement and there is no threat of acid spilling or any leakage whatsoever, and the fact that they are maintenance free meaning that you do not need to check and maintain the electrolyte level in the battery makes them all the more preferable, and being light in weight is also a plus.
Does a dry cell battery need water
Dry batteries do not need to add water.The “dry battery” of the non-rechargeable battery is a one-time use. After the power is exhausted, you cannot add water, because adding distilled water has no effect, and it cannot be charged, which is prone to danger.
A dry cell is a voltaic battery that uses some kind of absorbent, such as wood chips or gelatin, to make the contents into a paste that won’t spill. It is often used as a power source for flashlight lighting, radios, etc.
Advantages of a dry cell battery
- Dry cell battery are not expensive.
- The dry cell battery requires no maintenance.
- The dry cell battery has great resistance to cold weather.
- The battery can be used or placed in any position without fear of leakage or acid spilling.
- The small and compact size of a dry cell battery makes it suitable to power small electronic devices such as hearing aids, children’s toys, cameras, portable radios.
- Dry cell battery are available in different sizes and powers.
- They are safe and easy to transport over a long distance.
- Dry cell batterys can be placed in any position and fitted in a device without the risk of leakage.
Disadvantages of dry cell battery
- Risk of an explosion when exposed to too much heat due to the chemicals inside them.
- When not in use, it should be sealed and stored to prevent the moisture in the electrolyte from volatilizing.
- It cannot be recharged once all the power has been used up.
- For electrical appliances that are not used for a long time, the dry batteries should be removed to prevent the electrolyte from flowing out and corroding the electrical appliances.
Are dry cell battery still used
Dry cell battery are still being produced in great quantities and are used worldwide in devices and portable electronics such as children’s toys, mobile phones, laptops, flashlights, cameras, and portable radios.The standard working voltage for the dry cell battery is 1.5 volts, and the aa and aaa battery cell types are being used extensively worldwide.
What is the difference between a dry cell and a battery
A cell and battery are two different things, even though we sometimes use the terms interchangeably.
A cell consists of a chemical energy source which is then stored and converted to electrical energy, while a battery contains pre-supplied electrical energy from the factory it was produced from, and a battery can be charged and supplied electrical energy via an outlet.A battery stores electrical energy, whereas a cell generates energy by converting chemical energy into electrical energy.
Differences between the two:
- A cell is a unit (a pair of electrodes, the anode and cathode in an electrolyte) that converts chemical energy into electrical energy, while a battery is made up of a group of cells.
- A cell is either a wet or dry type, and it also includes a molten salt type, or the type of cell depends on the electrolytes used, while a battery is known to be either primary or secondary, which makes it rechargeable or non-rechargeable.
- A cell as a single unit is usually compact and light, while a battery is made up of several cells, thus making it bulky and weighty.
- A cell can supply power for a short period of time, while a battery which contains several cells can supply power for a long period of time and even go farther if it is a rechargeable battery type.
- Cells are cheap, while batteries cost more.
- Cells are majorly used for lighter tasks that require less energy, as seen in clocks and lamps, while batteries are used mostly for heavy-duty tasks and large equipment are majorly used in inverters, automobiles.
Is a lithium battery a dry cell
A Lithium metal primary battery is a dry cell made up of multiple dry cells because it stores energy over a long period of time and tends to lose power at a very slow rate, and it cannot be recharged once the power has been used up completely.Lithium batteries are primary batteries with metallic lithium as the anode, and they stand out from other primary batteries with their high charge density and high cost per unit.
Lithium batteries comprise many types of cathodes and electrolytes, but all have metallic lithium as their anode.Lithium battery as a primary dry cell battery means it cannot be recharged and must be disposed of after the electrical charge runs out. It has secondary batteries under it, and they include;
- Lithium-ion(Li-ion) Battery
The Li-ion battery is a rechargeable battery cell with a very high energy density and also stable. It is used in many consumer electronics and other devices. They are also used as backup power in telecommunications applications. The battery can store energy, and is used as portable energy in converters and power banks. The mainstream rechargeable battery on the market is lithium iron phosphate battery, this battery type offers safety and an excellent lifespan—ideal for emergency power supplies.
Here is a polymer lithium battery that is different from the cylindrical battery packaging method, this packaging method type battery makes use of a polymer electrolyte, and is majorly used where weight is a critical feature, this battery makes use of a polymer electrolyte, and is majorly used where weight is a critical feature, such as in some electric vehicles and radio-controlled aircraft.
Which lasts longer a dry cell battery or a lithium battery
The alkaline battery will be the dry cell battery used in comparison with the lithium battery
Lithium batteries last much longer because they have a high charge density which means they can hold more power and last longer than any other battery. Lithium batteries have a relatively high energy density, and in terms of charging and use, lithium batteries last longer and have a longer life, which makes them a good choice for high-tech devices.
Lithium batteries come in different sizes which include the aa, aaa, and 9V sizes.Lithium batteries can operate well in a wide temperature range, lasts up to 3x longer than any other battery type and are ideal for heavy use while being compact and lightweight for portable devices.
The lithium ion secondary battery possesses so many advantages
- The battery is known to have a long lifespan
- It is a rechargeable battery
- The lithium battery requires low maintenance
- It has a low self-discharge






























2 thoughts on “Are the non-rechargeable dry cell battery still in use”
For most up-to-date information you have to pay a visit world wide web and on internet I found this web site as a best web page
for newest updates.
This article presents clear idea in favor of the new visitors of blogging