Home » lithium ion battery knowledge » Extinguishing lithium ion battery fires – mainstream technologies and related research
Extinguishing lithium ion battery fires - mainstream technologies and related research
The proportion of new energy sources such as wind power and photovoltaics in the power system is gradually increasing. Due to the high volatility and randomness of new energy power generation, large-scale energy storage solutions needs to be deployed.
Lithium battery energy storage systems have developed rapidly in recent years due to their advantages such as high energy density, good economy, and short construction period.
However, fire and explosion accidents in lithium battery energy storage systems occur from time to time around the world, threatening the safe and stable operation of energy storage systems and restricting the development of battery energy storage systems. Extinguishing lithium ion battery fires is an international technology that urgently needs to be developed.

Difficulties in extinguishing lithium ion battery fires
Lithium battery energy storage system has risks on thermal runaway and fire in the whole life cycle, compared with traditional fire, extinguishing lithium ion battery fires mainly has two difficulties as below:
- Lithium battery fire is a composite combustion of gas and liquid, with the rapid diffusion and spread, megawatt-hour system fire source power is high, which makes the fast fire extinguishing difficult.
- The thermal runaway of lithium battery is an electrochemical reaction, which can be spontaneous and continuous, and it is difficult to prevent reignition after extinguishing lithium ion battery fires.
Therefore, the efficient technology of extinguishing lithium ion battery fires in energy storage system has become a hot topic of global researchers in recent years.
Based on the analysis of the research status, we will introduce the research on fluor-based rapid fire extinguishing and passivation cooling methods to prevent reignition, which can quickly extinguish the initial open fire of lithium energy storage battery.
And do not reignite for a long time after extinguishing lithium ion battery fires, and puts forward a new technical scheme to ensure the safety of lithium battery energy storage system.
Mainstream fire extinguishing technologies
Water extinguishing
Some countries, including the United States, use water injection fire pipes in the battery compartment to achieve the purpose of quickly extinguishing lithium ion battery fires through a large amount of external water injection.
However, the energy storage system use high voltage battery, water injection may lead to internal short circuit of the lithium-ion battery, resulting in large-scale damage to the equipment, and there is a risk of expanding the fire.
Water spray and water mist can quickly cool down and extinguish the fire, but in the energy storage system, the lithium battery module and cluster have a closed shell, and most of the water droplets cannot penetrate to the central flame area due to the shielding, resulting in reduced efficiency of extinguishing lithium ion battery fires and limited application.
In addition, water mist may produce CO, HF, H2 and other toxic and explosive gases during the process of extinguishing lithium ion battery fires, increasing the safety risk of water mist fire extinguishing.
Although water mist can be used as an effective method for extinguishing lithium ion battery fires, it may cause water pollution when mistakenly sprayed in application. The battery system voltage is high, and the water accumulation during water mist fire extinguishing may cause secondary damage such as short-circuit. Therefore, the battery system should be selected carefully according to the actual application requirements.
Gas extinguishing
The advantage of gas extinguishing is that it has strong insulation and can quickly extinguish open flames compared with water extinguishing.
At present, many lithium ion battery company use heptafluoropropane (C3HF7) as a gas extinguishing method in lithium battery energy storage systems, and the fire extinguishing mechanism includes physical inhibition and chemical inhibition, but physical inhibition is more powerful than chemical inhibition.
The physical heat absorption of sevofluoropropane can be divided into latent heat of gasification, specific heat and decomposition heat. For open flame of lithium battery, SEvofluoropropane can be quickly extinguished in a few seconds, but it is easy to reignite after the completion of injection.
Because the potential heat of sevofluoropropane is only 132kJ/kg, even though the open fire is extinguished, due to the failure to cool the lithium battery and stop the thermal runaway reaction, the internal chemical reaction of lithium battery still continues to produce heat, leading to reignition.
In addition, during the release process, heptafluoropropane will generate HF gas at high temperature, which may threaten the safety of rescue workers.
In recent years, a new type of clean gas extinguishing agent, perfluorohexanone (C6F12O), has begun to attract attention, and its fire extinguishing mechanism is mainly chemical inhibition, which can capture burning free radicals through fluorine-containing substances (CF3, CF2) and quickly extinguish the fire.
The main physical inhibition is the heat absorption of perfluorohexanone phase transition, which reduces the surface temperature of the battery. The latent heat of perfluorohexanone phase transition is 88kJ/kg, and the cooling performance is limited, but better than heptafluoropropane.
In order to improve the cooling ability of perfluorophenone in applications, intermittent spraying of perfluorophenone can improve the cooling efficiency, and the best effect is when the duty cycle is 54.3%.
In addition, it should be noted that when the volume fraction is below 2%, perfluorophenone will increase the battery temperature, possibly because the low concentration of perfluorophenone increases the flame speed and adiabatic temperature.
Moreover, the MFED1(toxicity index) of perfluorophenone after extinguishing the fire was higher, reaching 0.75. In order to achieve the coordination and unification of fire suppression and safety, it is recommended that the dosage of perfluorohexanone be 2.9g/(W·h) for extinguishing lithium ion battery fires.
The fluor-based fire extinguishing agent represented by perfluorohexanone has excellent fire extinguishing performance, but the cooling effect is not good enough and the amount is large and the cost is high. The cooling performance of fine water mist is strong, but it may cause external short circuit of the battery and expand the fire.
In addition, at present, extinguishing lithium ion battery fires is mainly concentrated in the single battery test research, and there is a lack of test verification for the application of large-scale energy storage system, and it is urgent to carry out a new high-efficiency fire extinguishing test research for the fire of large-size battery modules.
Fire extinguishing test research
Fire extinguishing methods
In view of the thermal runaway characteristics of energy storage lithium-ion batteries, researchers have proposed fluorine-based rapid fire extinguishing and passivation cooling methods to prevent re-ignition.
In the early stages of a battery fire, perfluorohexanone is used to quickly extinguish the open flame, and then low-pressure carbon dioxide is sprayed intermittently to cool down and prevent the battery fire from re-igniting. Low-pressure carbon dioxide is an excellent cooling medium at relatively low pressure (2.07MPa) and low temperature (-18~20℃).
It has the largest latent heat of vaporization (276.3kJ/kg) among existing mainstream gas fire extinguishing agents. Preliminary research found that low-pressure carbon dioxide has a strong heat-absorbing property due to its large heat of vaporization, and is effective in inhibiting thermal runaway of lithium batteries.
Experimental setup
The layout of the fire extinguishing test is as below.
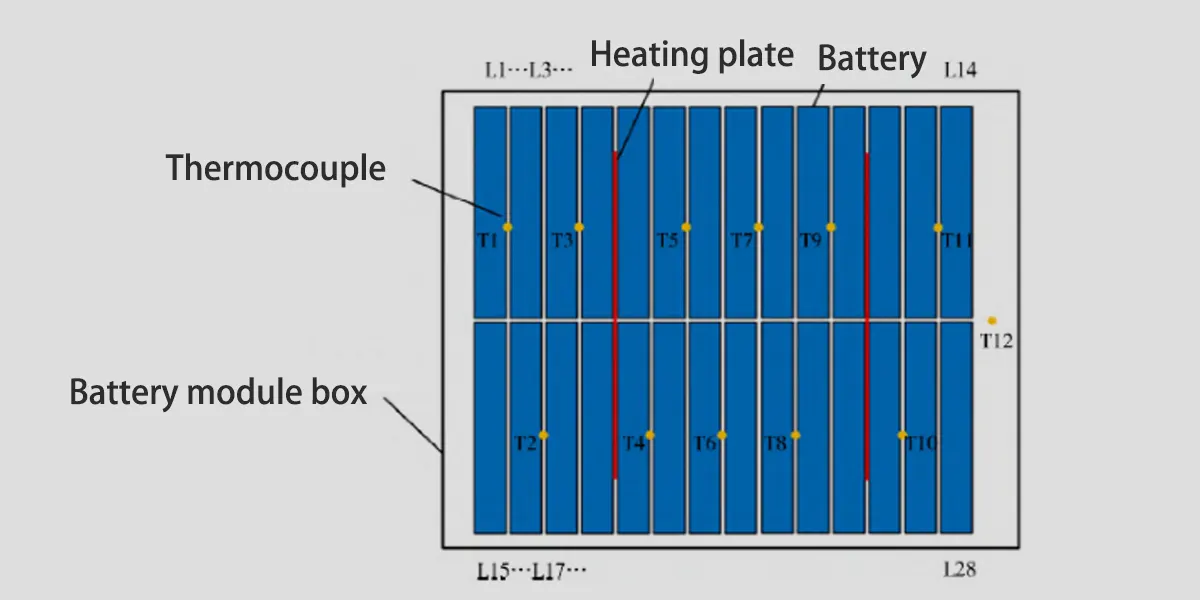
- Arrange a total of 28 72A·h/3.2V lithium iron phosphate batteries in the battery module box.
- Connect all batteries in series with copper bars to form a battery pack.
- Place two heating plates between the batteries and the power are both 1000W.
- At the same time, set a barbed wire mesh on the top of the battery module.
- While heating the battery, drop the barbed wire mesh on top of the battery module, causing an external short circuit.
- The coupling of heating and short circuit induces thermal runaway and fire in the battery.

Thermocouples T1-T11 are set between the batteries, as shown in the figure below, to collect surface temperature change data of the battery during thermal runaway and fire extinguishing. T12 measures the ambient temperature. Perfluorohexanone and low-pressure carbon dioxide fire extinguishing nozzles are installed directly above the battery box.
Test results
The image of the entire test process is as below. After the battery was continuously heated for about 35 minutes, the wire mesh was actively dropped on top of the battery module to achieve a short circuit between the cathode and anode poles. When the maximum temperature of the battery module cell surface reached 110°C, as shown in (b), a short circuit outside the battery produced a large number of sparks.
After 413 seconds, the battery caught fire, as shown in (c), and the maximum temperature of the battery core surface was 122°C. During the combustion process, a violently burning jet fire is formed, as shown in (d).
After continuing to burn for a while, start perfluorohexanone for extinguishing lithium ion battery fires, and at the same time turn off the power of the heating plate and stop heating. As shown in (e), the open flame is extinguished within 5 seconds, and then start low-pressure carbon dioxide intermittent cooling to inhibit battery re-ignition.

The test environment temperature was 30.5°C. When the battery pack is heated, the temperature rose linearly. When the measured battery surface temperature reached a maximum of 110°C, which is close to the critical temperature of thermal runaway, the battery module was externally short-circuited with a wire mesh.
But the battery did not catch fire immediately, and the temperature maintained its original trend and continued to rise until 150°C, when a short circuit produced an electric spark and caused the battery to catch fire.
The test used perfluorohexanone and low-pressure carbon dioxide for extinguishing lithium ion battery fires and preventing it from re-ignition. From the measurement results of T4 temperature, it showed that the open flame was quickly extinguished and the curve showed a steep downward trend. Subsequently, low-pressure carbon dioxide was sprayed intermittently to cool down a total of 4 times.
The test results showed that this fire-extinguishing medium can quickly reduce the ambient temperature, with the maximum cooling to -60°C. Since the short circuit has always existed and has not been removed, the local temperature of the battery pack still rises intermittently after the fire is extinguished.
For example, the T3 temperature increased around 3500s. This is mainly because the battery core was not heated evenly, but reached the critical thermal runaway temperature in stages. For example, T10 reached the critical surface temperature of 120°C first, and the battery L26 next to it cached fire first, and then the heat began to spread between the batteries.
In order to evaluate the cooling performance of low-pressure carbon dioxide injection on the battery, a typical temperature T4 was selected to calculate the cooling amount, cooling power and cooling efficiency of low-pressure carbon dioxide.
The calculation formula is as below:
Qc:Low-pressure carbon dioxide cooling capacity
A: Cross-sectional area of the module
λco2: Carbon dioxide thermal conductivity
d: Carbon dioxide nozzle hole diameter
ΔT: Temperature difference before and after low-pressure carbon dioxide injection
c: Battery specific heat capacity (1.1kJ/kg·℃)
m: Battery module mass (50.4kg)
qc: Cooling power
Δt: Low-pressure carbon dioxide injection time
e: Cooling efficiency
h: Latent heat of low pressure carbon dioxide
mc: Low-pressure carbon dioxide injection volume
| Low pressure carbon dioxide injection cooling performance parameters | ||||
|---|---|---|---|---|
| Injection times | Injection duration/s | Cooling capacity/kJ | Cooling power/W | Cooling efficiency/% |
| 1 | 36 | 1857.24 | 51. 59 | 44.80 |
| 2 | 26 | 1419.26 | 54.58 | 49.32 |
| 3 | 32 | 1829.52 | 57.17 | 51.54 |
| 4 | 30 | 1707.55 | 44.90 | 41.20 |
Compared with overcharging with battery charger, heating and other single modes that trigger battery thermal runaway fires, battery fires triggered by coupling failure modes of heating and short circuit are more difficult to control, mainly because the ignition source cannot be effectively eliminated, which also means the importance of continuously releasing low-cost and efficient cooling media.
This method can effectively slow down the intensity of the thermal runaway chain reaction, and can also exert a better inhibitory effect on severe thermal runaway such as an external short circuit of the battery.
The above experimental research can quickly extinguish the initial open fire of the energy storage lithium battery module and prevent it from re-igniting after the fire is extinguished. However, the technology of extinguishing lithium ion battery fires can only reduce fire losses afterwards, but cannot fundamentally solve the safety problem of battery fire and explosion after thermal runaway.
For this reason, it is more important to carry out in-depth research on intrinsically safe energy storage lithium-ion battery technology.